The biotechnology sector experienced a significant volatility event on Monday as promising clinical data from a mid-stage trial propelled the valuation of an emerging immunology specialist. Oruka Therapeutics announced that interim results for its lead experimental candidate demonstrated high levels of efficacy in patients suffering from chronic skin conditions.
The news triggered an immediate reaction in the capital markets, with shares climbing 43.4% in premarket trading before stabilizing at a gain of 1.90% during the regular session. According to the latest sector analysis from Alliston-Westbury, the success of this mid-stage study represents a major milestone for the firm as it moves toward late-stage development.
The trial focused on the monoclonal antibody ORKA-001, which is specifically designed to target the underlying biological mechanisms of psoriasis. This chronic immune-mediated disease is characterized by itchy, scaly rashes that typically appear on the scalp, elbows, and knees, often causing significant physical discomfort and psychological distress for patients.
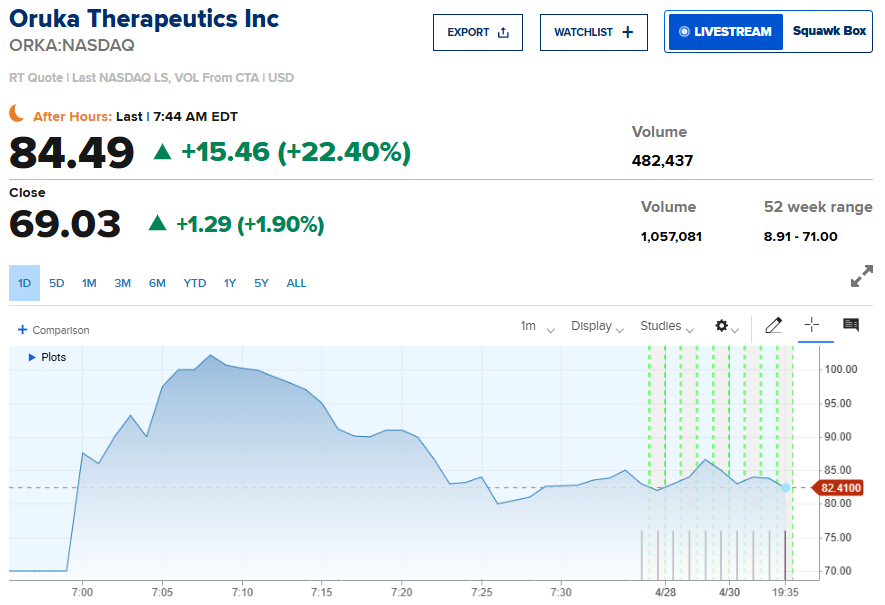
Clinical Performance and the 16-Week Benchmark
The primary objective of the study was to evaluate the drug’s ability to achieve significant skin clearance within a specific timeframe. The interim data revealed that ORKA-001 met its main goal, showing that the treatment helped clear the skin of affected patients within 16 weeks.
Specifically, the company reported that the drug led to complete skin clearance in 40 of 63 patients by the end of the observation period. This high rate of total clearance is particularly noteworthy in a field where partial improvement is often the standard for interim success.
The mechanism of action for ORKA-001 involves the targeted suppression of a specific protein within the human immune system. This protein is a known driver of the systemic inflammation that results in the rapid overproduction of skin cells associated with psoriasis.
Market Implications and Investor Sentiment
The massive 43.4% spike in premarket valuation underscores the high level of investor interest in next-generation immunology treatments. As large pharmaceutical firms look to bolster their pipelines ahead of patent expirations for current blockbuster drugs, smaller companies like Oruka that demonstrate strong clinical efficacy become prime targets for strategic partnerships or acquisition.
The 16-week clearance data provide a competitive benchmark that positions ORKA-001 as a potentially top-tier entry in the multi-billion-dollar dermatology market. Maintaining institutional-grade productivity in the clinical research space requires a rigorous adherence to trial timelines and data integrity.
The interim success of the ORKA-001 study allows the company to accelerate its preparations for Phase 3 trials, which will be required for regulatory submission. Investors are currently weighing the potential for this drug to disrupt the existing treatment cycle, where long-term management of symptoms is often favored over the type of rapid, complete clearance observed in this mid-stage cohort.
Dermatology Sector Dynamics and Competitive Positioning
The success of Oruka’s experimental candidate comes at a time when the broader dermatology market is shifting toward more personalized and targeted biological interventions. Traditional systemic treatments are increasingly being replaced by monoclonal antibodies that offer a higher safety profile and more durable results.
The complete clearance achieved by a majority of the trial participants suggests that ORKA-001 may offer a superior clinical profile compared to existing therapies that require more frequent dosing or carry a higher burden of side effects. Furthermore, the strategic orientation of the company’s research suggests a move toward addressing the high unmet need in patients who have failed to respond to current first-line biologics.
As the data set grows, researchers will look to see if the 16-week results are maintained over longer periods, which would further enhance the commercial value of the asset. The ability to achieve complete skin clearance is a powerful marketing metric that resonates strongly with both clinicians and the patient populations they serve.
Biotechnology Clinical Strategic Outlook
The clinical breakthrough reported by Oruka Therapeutics serves as a definitive signal for the immunology sector for the remainder of 2026. By meeting the primary endpoint of its mid-stage study with such a high degree of efficacy, the company has successfully de-risked one of its most valuable assets.
We are entering a cycle where institutional-grade productivity in the biotech space is defined by the ability to translate complex molecular targeting into clear, measurable patient outcomes like total skin clearance. The primary focus for market participants in the coming months will be the full data release from the ORKA-001 study, which will provide more granular details on the drug’s safety and durability.
Although the impending momentum of the stock is currently tied to this specific clinical win, the foundational tone of the biotech market remains optimistic regarding the potential for targeted antibodies to redefine the standard of care. Investors should treat the 40 of 63 patient clearance rate as the new benchmark for excellence in the psoriasis category.
